CBSE Class 10 Science Notes – Metals and Non-metals

1. Introduction
Metals are elements that are generally hard, lustrous, malleable, ductile, sonorous, and good conductors of heat and electricity. Examples include iron, copper, gold, and aluminium. Non-metals are elements that are generally soft, non-lustrous, brittle, and poor conductors of heat and electricity. Examples include carbon, sulphur, oxygen, and nitrogen.
- Metals: Used in jewellery (gold, silver), construction (iron), utensils (aluminium, stainless steel), electrical wires (copper)
- Non-metals: Essential for life (oxygen for breathing, carbon in organic compounds), used in fertilizers (nitrogen), water purification (chlorine)
- Board Exam Weightage: 6-8 marks (Theory + Practical)
- High-scoring: Reactivity series, extraction processes, ionic bonding
- Foundation for understanding materials and their applications
- NCERT Chapter 3: Metals and Non-metals (2024-25 Edition)
2. Chapter Overview
CBSE Class 10 Science Notes for this chapter cover:
- Physical properties of metals and non-metals
- Chemical properties (reaction with oxygen, water, acids, salts)
- Reactivity series of metals
- Extraction of metals (concentration, roasting, calcination, reduction)
- Corrosion and its prevention
- Ionic compounds and their properties
3. CBSE Class 10 Science Notes – Physical Properties
Physical Properties of Metals
| Property | Description | Examples |
|---|---|---|
| Malleable | Can be beaten into thin sheets | Aluminium foil, gold leaf |
| Ductile | Can be drawn into wires | Copper wires, gold wires |
| Good conductors | Conduct heat and electricity | Copper, aluminium, silver |
| Lustrous | Shiny surface | Gold, silver, copper |
| Sonorous | Produce ringing sound when struck | Bell metal, iron |
| High melting point | Generally solid at room temperature | Iron (1538°C), Copper (1085°C) |
| High density | Heavy for their size | Lead, gold, mercury |
Physical Properties of Non-metals
| Property | Description | Examples |
|---|---|---|
| Brittle | Break easily when hammered | Sulphur, phosphorus |
| Poor conductors | Do not conduct heat/electricity (except graphite) | Sulphur, phosphorus |
| Non-lustrous | Dull appearance (except iodine) | Carbon (coal), sulphur |
| Not sonorous | Do not produce ringing sound | All non-metals |
| Low melting point | Many are gases or soft solids | Oxygen, nitrogen, sulphur |
- Graphite (carbon): Non-metal that conducts electricity (used in pencils, electrodes)
- Mercury: Only metal that is liquid at room temperature
- Iodine: Non-metal that is lustrous (shiny black solid)
- Gallium and Caesium: Metals with very low melting points (melt on palm)
- Sodium and Potassium: Soft metals that can be cut with knife
4. CBSE Class 10 Science Notes – Chemical Properties
1. Reaction with Oxygen
Metals react with oxygen to form metal oxides. These are generally basic in nature, though some are amphoteric.
Amphoteric Oxides: Oxides that show both acidic and basic properties (react with both acids and bases)
Non-metals react with oxygen to form acidic or neutral oxides:
2. Reaction with Water
Different metals react differently with water based on their reactivity:
a) Reaction with cold water: Highly reactive metals (K, Na, Ca)
b) Reaction with hot water: Moderately reactive metals (Mg)
c) Reaction with steam: Less reactive metals (Zn, Fe, Al)
d) No reaction: Least reactive metals (Cu, Ag, Au)
3. Reaction with Acids
Metals above hydrogen in reactivity series displace hydrogen from dilute acids.
- Nitric acid (HNO₃) is a strong oxidizing agent, so it generally does not liberate hydrogen gas with metals
- Copper, silver, and gold do not react with dilute acids as they are below hydrogen in reactivity series
- Aluminium and iron react slowly due to formation of protective oxide layer
4. Displacement Reactions
A more reactive metal can displace a less reactive metal from its salt solution.
Observation: Blue solution becomes colorless, reddish-brown copper deposits on zinc
Observation: Blue solution turns light green, copper deposited
Observation: Colorless solution turns blue, silver-white deposit on copper
5. CBSE Class 10 Science Notes – Reactivity Series & Extraction
Reactivity Series of Metals
The reactivity series is a list of metals arranged in order of decreasing reactivity. Metals at the top are most reactive, while those at the bottom are least reactive.
K > Na > Ca > Mg > Al > Zn > Fe > Pb > H > Cu > Hg > Ag > Au
Mnemonic: "Please Stop Calling Me A Zebra Instead Try Learning How Copper Saves Gold"
- Potassium
- Sodium
- Calcium
- Magnesium
- Aluminium
- Zinc
- Iron
- Tin
- Lead
- Hydrogen
- Copper
- Silver
- Gold
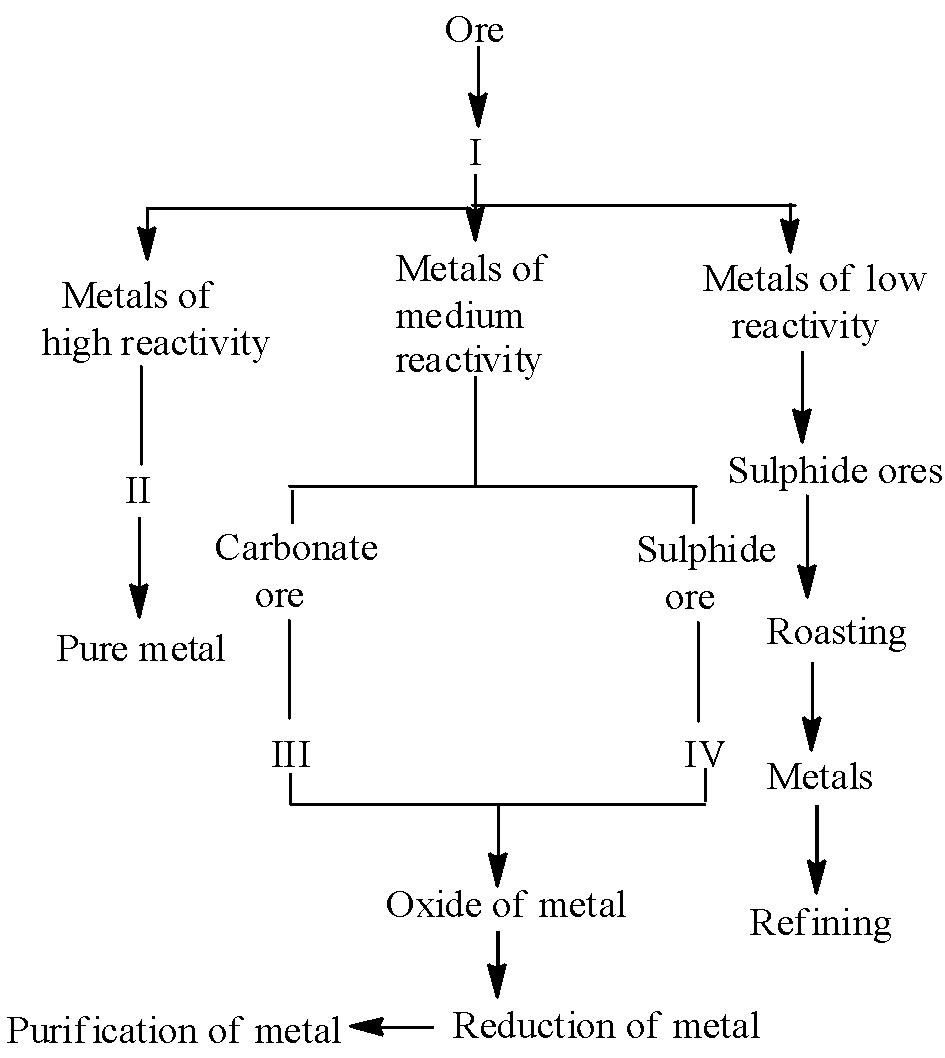
Extraction of Metals
The process of obtaining metals from their ores and refining them is called metallurgy. The method of extraction depends on the position of metal in reactivity series.
Steps in Extraction of Metals (Middle Reactivity):
Step 1: Concentration of Ore
Removal of impurities (gangue) from ore to increase metal content.
- Hydraulic washing: Based on gravity difference (for heavy ores like iron)
- Froth flotation: For sulphide ores (ZnS, PbS)
- Magnetic separation: For magnetic ores (magnetite Fe₃O₄)
Step 2: Conversion to Metal Oxide
| Process | Definition | Used For | Example |
|---|---|---|---|
| Roasting | Heating sulphide ore in excess of air | Sulphide ores | 2ZnS + 3O₂ → 2ZnO + 2SO₂↑ |
| Calcination | Heating carbonate ore in limited air | Carbonate ores | ZnCO₃ → ZnO + CO₂↑ |
Step 3: Reduction to Metal
Metal oxide is reduced to metal using suitable reducing agent:
- Carbon (coke): For Zn, Fe, Pb
- Carbon monoxide: For iron (in blast furnace)
- Aluminium (Thermite process): For Cr, Mn (high melting point metals)
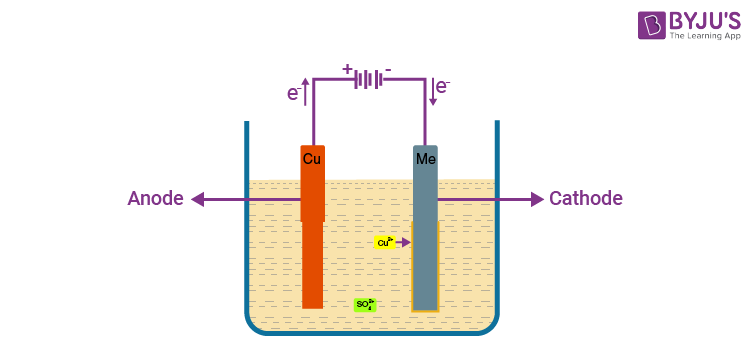
Step 4: Refining
Purification of extracted metal using electrolytic refining.
- Anode: Impure metal
- Cathode: Pure metal strip
- Electrolyte: Solution of metal salt
On passing current, pure metal from anode dissolves and deposits on cathode. Impurities settle as anode mud.
6. CBSE Class 10 Science Notes – Ionic Compounds
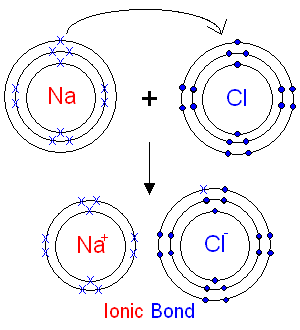
Formation of Ionic Bond
Ionic compounds are formed by transfer of electrons from metals to non-metals. Metals lose electrons to form positive ions (cations), while non-metals gain electrons to form negative ions (anions). The electrostatic attraction between these oppositely charged ions forms ionic bond.
Example: Formation of Sodium Chloride (NaCl)
- Sodium (Na): Atomic number 11, electronic configuration 2,8,1. Loses 1 electron to achieve octet → Na⁺
- Chlorine (Cl): Atomic number 17, electronic configuration 2,8,7. Gains 1 electron to achieve octet → Cl⁻
Properties of Ionic Compounds
| Property | Explanation |
|---|---|
| High melting and boiling points | Strong electrostatic forces between ions require lot of energy to break |
| Solid state | Exist as hard solids at room temperature |
| Brittle | Break into pieces when pressure applied (ions shift, like charges repel) |
| Soluble in water | Water molecules separate the ions (hydration) |
| Conduct electricity in molten/aqueous state | Ions are free to move and carry current |
| Insulators in solid state | Ions are fixed in position, cannot move |
7. Important Points (Quick Revision)
- Reactivity Series: K > Na > Ca > Mg > Al > Zn > Fe > Pb > H > Cu > Hg > Ag > Au
- Roasting: Sulphide ore + excess air → Metal oxide + SO₂
- Calcination: Carbonate ore + limited air → Metal oxide + CO₂
- Ionic compounds: High melting point, soluble in water, conduct when molten/aqueous
- Corrosion prevention: Painting, galvanization, oiling, alloying, electroplating
- Displacement rule: More reactive metal displaces less reactive from salt solution
- Amphoteric oxides: React with both acids and bases (Al₂O₃, ZnO)
8. Solved Examples (CBSE Pattern)
Example 1: Identify Metal/Non-metal (2 Marks)
Question: An element X forms an oxide X₂O₃ which is acidic in nature. Identify whether X is a metal or non-metal. Name another element that forms similar oxide.
Solution:
- Since the oxide is acidic, X is a non-metal
- Non-metals form acidic oxides
- Examples: Sulphur forms SO₂/SO₃, Carbon forms CO₂, Nitrogen forms NO₂
Answer: X is a non-metal. Another example is sulphur (S) forming SO₂.
Example 2: Reaction Type Question (2 Marks)
Question: A student took the following metals: Sodium, Magnesium, Copper. Arrange them in decreasing order of reactivity with water.
Solution:
- Sodium: Reacts vigorously with cold water
- Magnesium: Reacts with hot water/steam
- Copper: Does not react with water
Answer: Sodium > Magnesium > Copper
Example 3: Reactivity Comparison (3 Marks)
Question: Metal X displaces metal Y from Y₂(SO₄)₃ solution. Metal Y displaces Zn from ZnSO₄ solution. Arrange X, Y, Zn in decreasing order of reactivity. Why can't X displace Zn from ZnSO₄?
Solution:
- X displaces Y → X is more reactive than Y
- Y displaces Zn → Y is more reactive than Zn
- Therefore: X > Y > Zn
X CAN displace Zn from ZnSO₄ because X is more reactive than Zn. The question statement might have an error, OR if X cannot displace Zn, then the order would be different. Based on given information, X is most reactive.
Example 4: Extraction Reasoning (3 Marks)
Question: Why is sodium metal not extracted from its oxide by reduction with carbon?
Answer:
- Sodium is a highly reactive metal (at top of reactivity series)
- It has strong affinity for oxygen
- Carbon cannot reduce sodium oxide because carbon is less reactive than sodium
- Sodium is extracted by electrolysis of molten NaCl (not by reduction)
Example 5: 5-Mark Conceptual Question
Question: a) Give the steps involved in the extraction of metals of middle reactivity from their sulphide ores.
b) Differentiate between roasting and calcination with chemical equations.
c) Why are carbonate and sulphide ores converted to oxides before reduction?
a) Steps for extraction from sulphide ores:
- Concentration: Froth flotation method
- Roasting: Convert sulphide to oxide
- Reduction: Heat oxide with reducing agent (carbon)
- Refining: Electrolytic refining
b) Difference:
| Roasting | Calcination |
|---|---|
| Sulphide ore heated in excess air | Carbonate ore heated in limited air |
| 2ZnS + 3O₂ → 2ZnO + 2SO₂ | ZnCO₃ → ZnO + CO₂ |
c) It is easier to reduce metal oxides to metals than to reduce sulphides or carbonates directly. Oxides give better yield and purer metal.
Example 6: Case-Study Based Question
Passage: Three students A, B, and C were given three unknown metals X, Y, and Z. They performed the following experiments:
- Metal X reacted vigorously with cold water
- Metal Y reacted with steam but not cold water
- Metal Z did not react with water or dilute acid
Questions: a) Arrange X, Y, Z in decreasing order of reactivity. b) Identify possible metals. c) Which metal can be used for making cooking utensils? Why?
a) Reactivity order: X > Y > Z
b) Identification:
- X could be Sodium or Potassium (reacts with cold water)
- Y could be Iron or Zinc (reacts with steam)
- Z could be Copper or Silver (no reaction)
c) Y (Iron or Aluminium) can be used for cooking utensils because:
- Moderately reactive, does not react vigorously with water
- Good conductor of heat
- Forms protective oxide layer (especially aluminium)
9. Smart Tricks & Memory Aids
"Please Stop Calling Me A Zebra Instead Try Learning How Copper Saves Gold"
Or alternative: "King Narayan Can Make A Zoo In London Having Common Sense Generally"
- K - Potassium
- N - Sodium
- C - Calcium
- M - Magnesium
- A - Aluminium
- Z - Zinc
- I - Iron
- L - Lead
- H - Hydrogen
- C - Copper
- S - Silver
- G - Gold
Concentration → Roasting/Calcination → Reduction → Refining
Remember: "CRRR" or "Chemistry Requires Regular Revision"
- Metal + Non-metal → Ionic compound (NaCl, MgO)
- High melting point → Ionic
- Soluble in water → Usually ionic
- Conducts electricity when molten → Ionic
- Roasting → Rich in air (excess air) → Sulphide ores
- Calcination → Controlled air (limited air) → Carbonate ores
- Roasting produces SO₂, Calcination produces CO₂
- Always write balanced chemical equations with state symbols
- For reactivity questions: Refer to reactivity series
- For extraction: Mention four steps clearly
- For ionic compounds: Explain electron transfer mechanism
- For corrosion: Must mention both air and moisture
- Remember exceptions (graphite, mercury, iodine, gallium)
10. Visual Learning – Diagrams

11. Most Important Board Questions
1 Mark Questions
2-3 Mark Questions
4-5 Mark Questions
12. Common Mistakes to Avoid
| Mistake | Correction |
|---|---|
| Confusing roasting and calcination | Roasting = excess air, sulphide ore; Calcination = limited air, carbonate ore |
| Wrong reactivity order | Remember: K Na Ca Mg Al Zn Fe Pb H Cu Hg Ag Au |
| Incorrect equation balancing | Always check atom count on both sides |
| Misunderstanding ionic bonding | It's electron TRANSFER, not sharing (that's covalent) |
| Forgetting exceptions | Graphite conducts, mercury is liquid, iodine is lustrous |
13. Practice Section
Multiple Choice Questions (MCQs)
Q1. Which metal is liquid at room temperature?
- Iron
- Aluminium
- Mercury
- Silver
Q2. The process of heating sulphide ore in excess air is called:
- Calcination
- Roasting
- Smelting
- Refining
Q3. Which of the following is an ionic compound?
- HCl
- CO₂
- NaCl
- CH₄
Assertion-Reason Question
Assertion (A): Sodium is stored under kerosene oil.
Reason (R): Sodium reacts with moisture in air.
Answer: Both A and R are true and R is the correct explanation of A.
Explanation: Sodium is highly reactive and reacts with moisture (and oxygen) in air. Kerosene prevents this contact.
14. Frequently Asked Questions (FAQ)
15. Conclusion
Master Metals and Non-metals for Board Success!
These comprehensive CBSE Class 10 Notes for "Metals and Non-metals" cover all essential concepts, reactivity series, extraction processes, and solved examples to help you excel in your board examinations.
Key Takeaways:
- Master the reactivity series and its applications
- Understand extraction steps: Concentration → Roasting/Calcination → Reduction → Refining
- Remember properties of ionic compounds
- Practice writing balanced chemical equations
- Know the exceptions (graphite, mercury, iodine)
Next Chapter: Carbon and its Compounds – CBSE Class 10 Notes
Best of luck for your CBSE Class 10 Board Examinations!
These CBSE Class 10 Science Notes are prepared by experienced Chemistry faculty following the latest NCERT curriculum.

