CBSE Class 10 Science Notes – Chemical Reactions and Equations

1. Introduction
A chemical reaction is a process in which one or more substances (reactants) are converted into one or more new substances (products) with different chemical properties. This involves breaking and forming of chemical bonds.
- Rusting of iron: Iron reacts with oxygen and moisture to form hydrated iron oxide (rust)
- Burning of fuel: Combustion of wood, LPG, petrol produces heat and light
- Digestion of food: Complex food molecules break down into simpler substances
- Photosynthesis: Plants convert CO₂ and water into glucose using sunlight
- Baking of cake: Heat causes chemical changes in ingredients
- Board Exam Weightage: 6-8 marks (Theory + Practical)
- Foundation for entire Chemistry syllabus
- High-scoring: Balancing equations, identifying reaction types, redox concepts
- NCERT Chapter 1: Chemical Reactions and Equations (2024-25 Edition)
2. Chapter Overview
CBSE Class 10 Science Notes for this chapter cover:
- Writing and balancing chemical equations
- Law of Conservation of Mass
- Types of chemical reactions (Combination, Decomposition, Displacement, Double Displacement)
- Oxidation and Reduction (Redox reactions)
- Corrosion of metals (Rusting of iron)
- Rancidity and its prevention
3. CBSE Class 10 Science Notes – Writing & Balancing Chemical Equations
What is a Chemical Equation?
A chemical equation is a symbolic representation of a chemical reaction using chemical formulas. The reactants are written on the left side and products on the right side, with an arrow (→) pointing from reactants to products.
Types of Chemical Equations
1. Word Equation: Uses words to describe reactants and products
2. Skeletal (Unbalanced) Equation: Uses chemical formulas but not balanced
3. Balanced Chemical Equation: Has equal number of atoms of each element on both sides
Law of Conservation of Mass
Proposed by Antoine Lavoisier (1789), this law states that mass can neither be created nor destroyed in a chemical reaction. The total mass of reactants equals the total mass of products. This is why we balance chemical equations.
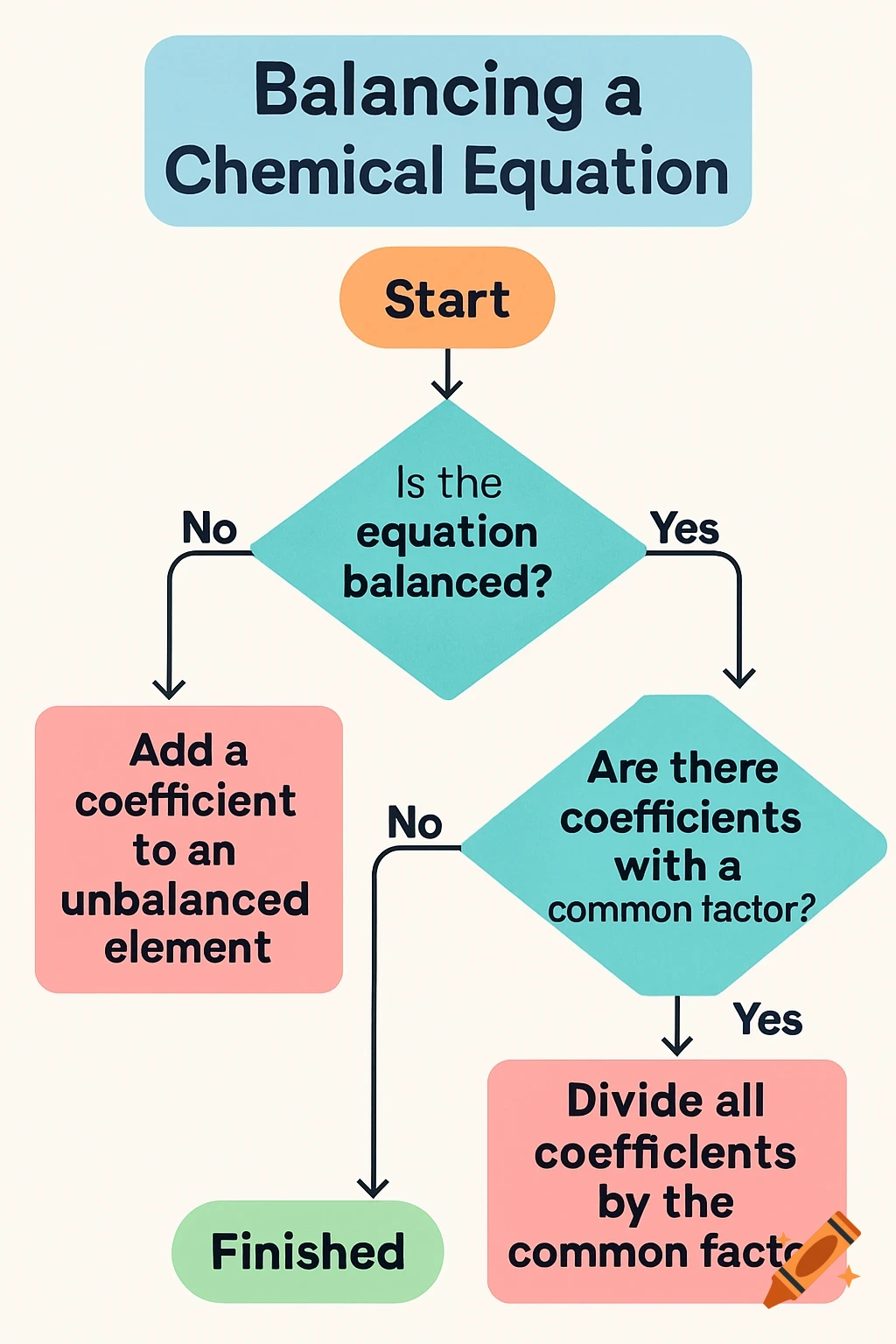
Balancing Chemical Equations - Hit and Trial Method
- Write the word equation and then skeletal equation using correct formulas
- Count atoms of each element on both sides
- Balance one element at a time by adding coefficients (start with metals, then non-metals, then hydrogen, finally oxygen)
- Never change subscripts in formulas - only add coefficients before formulas
- Make coefficients simplest whole numbers
- Verify atom count on both sides
Example: Balance the equation Fe + H₂O → Fe₃O₄ + H₂
Step 1: Count atoms
- Left side: Fe = 1, H = 2, O = 1
- Right side: Fe = 3, H = 2, O = 4
Step 2: Balance Fe atoms - multiply Fe by 3
Step 3: Balance O atoms - multiply H₂O by 4
Step 4: Balance H atoms - multiply H₂ by 4
Step 5: Verify - Left: Fe=3, H=8, O=4 | Right: Fe=3, H=8, O=4 ✓
State Symbols (Important for Board Exams)
| Symbol | State | Example |
|---|---|---|
| (s) | Solid | NaCl(s), CaCO₃(s) |
| (l) | Liquid | H₂O(l), H₂SO₄(l) |
| (g) | Gas | H₂(g), O₂(g), CO₂(g) |
| (aq) | Aqueous (dissolved in water) | NaCl(aq), HCl(aq) |
4. CBSE Class 10 Science Notes – Types of Chemical Reactions
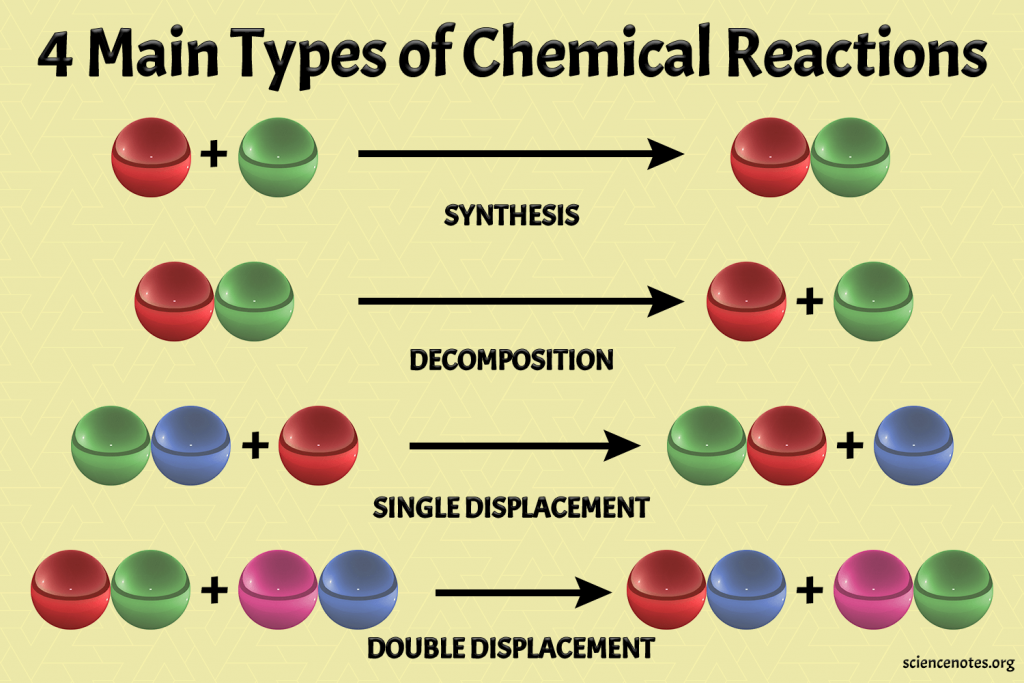
1. Combination Reaction
Two or more reactants combine to form a single product. Usually exothermic (releases heat).
Examples:
2. Decomposition Reaction
A single compound breaks down into two or more simpler substances. Usually requires energy (endothermic).
Types of Decomposition:
a) Thermal Decomposition (by heat):
Observation: Green crystals change to reddish-brown solid
b) Electrolytic Decomposition (by electricity):
c) Photochemical Decomposition (by light):
3. Displacement Reaction
A more reactive element displaces a less reactive element from its compound.
Examples:
Observation: Blue solution becomes colorless, reddish-brown copper deposits
K > Na > Ca > Mg > Al > Zn > Fe > Pb > H > Cu > Ag > Au
A metal can displace any metal below it in the series from its salt solution.
4. Double Displacement Reaction
Two compounds exchange ions to form two new compounds.
Examples:
5. Precipitation Reaction
A type of double displacement where an insoluble solid (precipitate) is formed.
Observation: Yellow precipitate of lead iodide
Comparison Table: Types of Reactions
| Type | General Form | Key Feature | Example |
|---|---|---|---|
| Combination | A + B → AB | Two or more → One | 2H₂ + O₂ → 2H₂O |
| Decomposition | AB → A + B | One → Two or more | 2H₂O₂ → 2H₂O + O₂ |
| Displacement | A + BC → AC + B | More reactive displaces less reactive | Zn + CuSO₄ → ZnSO₄ + Cu |
| Double Displacement | AB + CD → AD + CB | Ion exchange between two compounds | Na₂SO₄ + BaCl₂ → BaSO₄ + 2NaCl |
5. CBSE Class 10 Science Notes – Oxidation & Reduction (Redox)
Oxidation
A chemical process involving:
- Addition of oxygen
- Removal of hydrogen
- Loss of electrons
Reduction
A chemical process involving:
- Removal of oxygen
- Addition of hydrogen
- Gain of electrons
Redox Reaction
A reaction where oxidation and reduction occur simultaneously. One substance is oxidized while another is reduced.
- ZnO is reduced to Zn (oxygen removed)
- C is oxidized to CO (oxygen added)
Oxidizing and Reducing Agents
| Agent | Action | Itself Gets | Example |
|---|---|---|---|
| Oxidizing Agent | Oxidizes other substance | Reduced | O₂, KMnO₄, CuO |
| Reducing Agent | Reduces other substance | Oxidized | H₂, C, CO |
OIL RIG - Oxidation Is Loss (of electrons), Reduction Is Gain (of electrons)
Or: LEO says GER - Lose Electrons Oxidation, Gain Electrons Reduction
6. CBSE Class 10 Science Notes – Corrosion & Rancidity
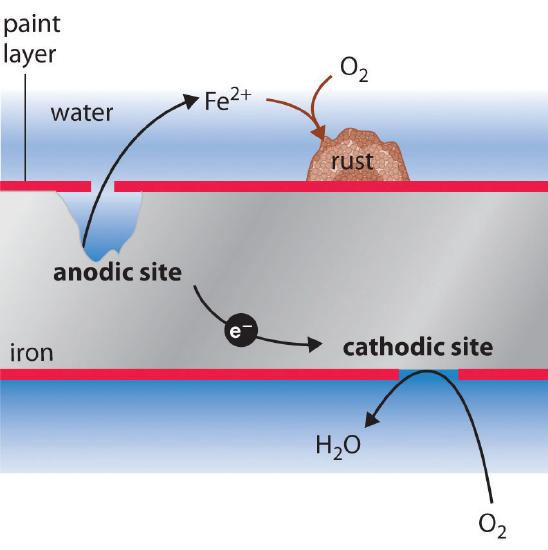
Corrosion
The process of deterioration of metals due to chemical attack by moisture, acids, gases, etc., forming undesirable compounds (oxides, carbonates, sulphides) on the surface.
Rusting of Iron
Conditions required: Both oxygen and moisture (water) must be present
Characteristics of Rust:
- Soft, porous, reddish-brown substance
- Not attached firmly to iron surface
- Weak and brittle
- Causes gradual destruction of iron articles
Prevention of Rusting
- Painting: Creates protective barrier (gates, railings)
- Oiling/Greasing: Prevents contact with air and moisture (machinery)
- Galvanization: Coating with zinc (roofing sheets, buckets)
- Electroplating: Coating with chromium or nickel
- Alloying: Making stainless steel (iron + chromium + nickel)
- Enamel coating: Glass-like coating (cooking utensils)
Rancidity
The oxidation of fats and oils in food materials when exposed to air, resulting in unpleasant smell and taste. This makes food unfit for consumption.
Prevention of Rancidity
- Adding antioxidants: BHA (Butylated Hydroxyanisole), BHT
- Refrigeration: Slows down oxidation
- Airtight containers: Prevents exposure to air
- Flushing with nitrogen: Inert gas prevents oxidation (chip packets)
- Storing away from light: Light accelerates oxidation
7. Important Points (Quick Revision)
- Law of Conservation of Mass: Mass is neither created nor destroyed; atoms are rearranged
- Balancing: Only coefficients can be changed, never subscripts
- Combination: Multiple reactants → Single product (Exothermic)
- Decomposition: Single reactant → Multiple products (Endothermic)
- Displacement: More reactive metal displaces less reactive from salt solution
- Double Displacement: Ion exchange; precipitation if insoluble product forms
- Redox: Oxidation and reduction occur simultaneously
- Rusting: Requires both O₂ and H₂O; prevention by barrier methods
- Rancidity: Oxidation of fats; prevented by antioxidants and nitrogen flushing
8. Solved Examples (CBSE Pattern)
Example 1: Balancing Chemical Equation (2 Marks)
Question: Balance the following chemical equation: Al + HCl → AlCl₃ + H₂
Step-by-step Solution:
- Count atoms: Left - Al=1, H=1, Cl=1 | Right - Al=1, H=2, Cl=3
- Balance Cl: Multiply HCl by 3 → Al + 3HCl → AlCl₃ + H₂
- Balance H: Multiply H₂ by 3/2 → Al + 3HCl → AlCl₃ + 3/2H₂
- Remove fraction: Multiply entire equation by 2
Balanced Equation:
Example 2: Identify Reaction Type (2 Marks)
Question: Identify the type of reaction in each case:
a) 2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂
b) Zn + CuSO₄ → ZnSO₄ + Cu
a) Decomposition Reaction - A single compound breaks down into multiple simpler substances upon heating.
b) Displacement Reaction - Zinc (more reactive) displaces copper (less reactive) from copper sulphate solution.
Example 3: Redox Reaction Identification (3 Marks)
Question: In the reaction CuO + H₂ → Cu + H₂O, identify:
a) Substance oxidized
b) Substance reduced
c) Oxidizing agent
d) Reducing agent
Analysis:
- CuO loses oxygen → becomes Cu (Reduction)
- H₂ gains oxygen → becomes H₂O (Oxidation)
Answers:
- a) H₂ is oxidized (gains oxygen)
- b) CuO is reduced (loses oxygen)
- c) CuO is oxidizing agent (oxidizes H₂, itself gets reduced)
- d) H₂ is reducing agent (reduces CuO, itself gets oxidized)
Example 4: Corrosion Reasoning (3 Marks)
Question: Why do iron nails rust faster in coastal areas than in deserts? Suggest two methods to prevent rusting.
Explanation: Rusting requires both oxygen and moisture. Coastal areas have high humidity (moisture in air) and salty air accelerates corrosion. Deserts have dry air with very low moisture.
Prevention Methods:
- Painting - creates protective barrier
- Galvanization - coating with zinc
- Oiling/Greasing - prevents air contact
- Electroplating - with chromium/nickel
Example 5: 5-Mark Conceptual Question
Question: a) What is a balanced chemical equation? Why should it be balanced?
b) Write balanced equations for:
i) Phosphorus burns in chlorine to form phosphorus pentachloride
ii) Sodium hydroxide reacts with hydrochloric acid
iii) Zinc carbonate is heated
a) Balanced Chemical Equation: An equation with equal number of atoms of each element on both sides. It must be balanced according to Law of Conservation of Mass - mass can neither be created nor destroyed.
b) Balanced Equations:
Example 6: Case-Study Based Question
Passage: A student observed that a shiny brown element 'X' when heated in air turned black. When hydrogen gas was passed over the heated black substance, it again turned shiny brown.
Questions: a) Name element 'X' and black compound. b) Write chemical equations. c) Identify types of reactions.
a) Element 'X' is Copper (Cu). Black compound is Copper(II) oxide (CuO).
b) Equations:
c) Reaction Types:
- First: Combination reaction
- Second: Redox reaction (Displacement + Reduction)
9. Smart Tricks & Memory Aids
Remember order: Metals → Non-metals → Hydrogen → Oxygen
Mnemonic: "Mama Never Hits Oliver"
Always balance oxygen last as it appears in multiple compounds.
- Combination: Many → One (Marriage - two become one)
- Decomposition: One → Many (Divorce - one becomes many)
- Displacement: One kicks out another (Bullying - strong displaces weak)
- Double Displacement: Partners swap (Exchange dance)
OIL RIG: Oxidation Is Loss (of electrons), Reduction Is Gain (of electrons)
Or: LEO the lion says GER
Lose Electrons = Oxidation, Gain Electrons = Reduction
- (s) - Solid
- (l) - Liquid
- (g) - Gas
- (aq) - Aqueous (aqua = water)
- Always write word equation first, then balanced chemical equation
- Include state symbols for full marks
- For reaction type questions, write definition + equation
- In redox questions, clearly mention what gains/loses oxygen
- For corrosion: Must mention both O₂ and H₂O required
- Write conditions (heat, light, electricity) above the arrow
10. Visual Learning – Diagrams


11. Most Important Board Questions
1 Mark Questions
2-3 Mark Questions
4-5 Mark Questions
12. Common Mistakes to Avoid
| Mistake | Correction |
|---|---|
| Changing subscripts while balancing | Only change coefficients, never subscripts in formulas |
| Wrong reaction type identification | Count reactants and products carefully |
| Confusing oxidation and reduction | Oxidation = gain of O/loss of H; Reduction = loss of O/gain of H |
| Ignoring state symbols | Always include (s), (l), (g), (aq) for full marks |
| Incomplete rusting explanation | Must mention both oxygen AND moisture required |
| Wrong reactivity series order | Remember: K Na Ca Mg Al Zn Fe Pb H Cu Ag Au |
13. Practice Section
Multiple Choice Questions (MCQs)
Q1. Which of the following is a displacement reaction?
- 2H₂ + O₂ → 2H₂O
- Zn + CuSO₄ → ZnSO₄ + Cu
- CaCO₃ → CaO + CO₂
- NaOH + HCl → NaCl + H₂O
Q2. The reaction 2AgCl → 2Ag + Cl₂ is an example of:
- Thermal decomposition
- Photochemical decomposition
- Electrolytic decomposition
- Displacement reaction
Q3. In the reaction CuO + H₂ → Cu + H₂O, CuO acts as:
- Reducing agent
- Oxidizing agent
- Catalyst
- Inert substance
Assertion-Reason Question
Assertion (A): White silver chloride turns grey in sunlight.
Reason (R): Silver chloride undergoes decomposition reaction in presence of sunlight.
Answer: Both A and R are true and R is the correct explanation of A.
Explanation: 2AgCl → 2Ag + Cl₂. Silver formed is grey, chlorine escapes.
14. Frequently Asked Questions (FAQ)
15. Conclusion
Master Chemical Reactions for Board Success!
These comprehensive CBSE Class 10 Notes for "Chemical Reactions and Equations" cover all essential concepts, balanced equations, and solved examples to help you excel in your board examinations.
Key Takeaways:
- Practice balancing equations using Hit and Trial method
- Remember state symbols (s, l, g, aq) for full marks
- Identify reaction types by counting reactants and products
- Understand redox: OIL RIG (Oxidation Is Loss, Reduction Is Gain)
- Corrosion needs O₂ + H₂O; Rancidity is oxidation of fats
Next Chapter: Acids, Bases and Salts – CBSE Class 10 Notes
Best of luck for your CBSE Class 10 Board Examinations!
These CBSE Class 10 Science Notes are prepared by experienced Chemistry faculty following the latest NCERT curriculum.

