CBSE Class 10 Science Notes – Acids, Bases and Salts

1. Introduction
Acids are substances that taste sour, turn blue litmus red, and produce hydrogen ions (H⁺) in aqueous solution. Bases taste bitter, feel soapy, turn red litmus blue, and produce hydroxide ions (OH⁻) in solution. Salts are ionic compounds formed by neutralization of acids and bases.
- Acids: Lemon (citric acid), vinegar (acetic acid), curd (lactic acid), tamarind (tartaric acid)
- Bases: Soap (sodium/potassium salts), baking soda (sodium bicarbonate), lime water (calcium hydroxide)
- Salts: Common salt (NaCl), baking powder, bleaching powder
- Board Exam Weightage: 6-8 marks (Theory + Practical)
- High-scoring: pH scale, chemical reactions, salt preparation equations
- Foundation for understanding chemical behavior
- NCERT Chapter 2: Acids, Bases and Salts (2024-25 Edition)
2. Chapter Overview
CBSE Class 10 Science Notes for this chapter cover:
- Properties of acids and bases
- Chemical reactions of acids and bases (with metals, carbonates, each other)
- Strength of acids and bases (concentrated vs dilute, strong vs weak)
- pH scale and its importance in daily life
- Salts and their family (acidic, basic, neutral salts)
- Chemicals from common salt (Chlor-alkali process)
- Preparation and uses of washing soda and baking soda
3. CBSE Class 10 Science Notes – Properties of Acids & Bases
Acids
Acids are substances that produce hydrogen ions (H⁺) or hydronium ions (H₃O⁺) when dissolved in water.
Physical Properties of Acids
- Sour taste
- Turn blue litmus paper red
- Conduct electricity in aqueous solution (electrolytes)
- pH less than 7
Chemical Properties of Acids
1. Reaction with Metals: Acids react with active metals to produce salt and hydrogen gas
Test for H₂ gas: Burning candle brought near gas produces 'pop' sound
2. Reaction with Metal Carbonates and Hydrogen Carbonates: Produce salt, water and carbon dioxide gas
Test for CO₂ gas: Turns lime water milky (Ca(OH)₂ + CO₂ → CaCO₃↓ + H₂O)
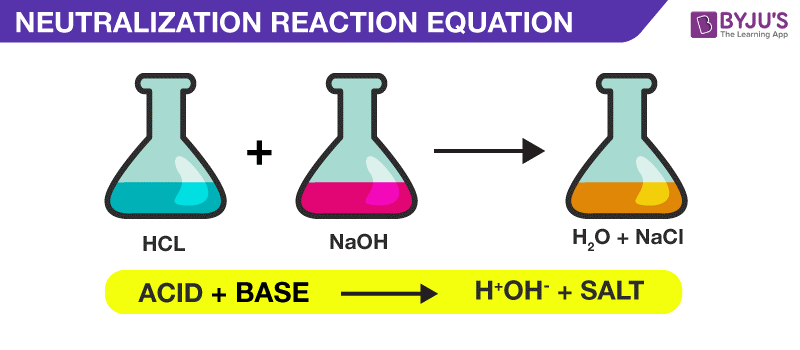
3. Reaction with Metal Oxides: Produce salt and water (neutralization)
4. Reaction with Bases: Neutralization reaction (see Section 4)
Bases
Bases are substances that produce hydroxide ions (OH⁻) when dissolved in water.
Physical Properties of Bases
- Bitter taste
- Soapy touch
- Turn red litmus paper blue
- Conduct electricity in aqueous solution
- pH greater than 7
Chemical Properties of Bases
1. Reaction with Metals: Certain metals (Zn, Al) react with strong bases to produce salt and hydrogen
(Sodium zincate)
(Sodium aluminate)
2. Reaction with Non-metal Oxides: Produce salt and water
3. Reaction with Acids: Neutralization reaction
| Property | Acids | Bases |
|---|---|---|
| Ions produced | H⁺ / H₃O⁺ | OH⁻ |
| Taste | Sour | Bitter |
| Touch | Not soapy | Soapy |
| Litmus test | Blue → Red | Red → Blue |
| pH | < 7 | > 7 |
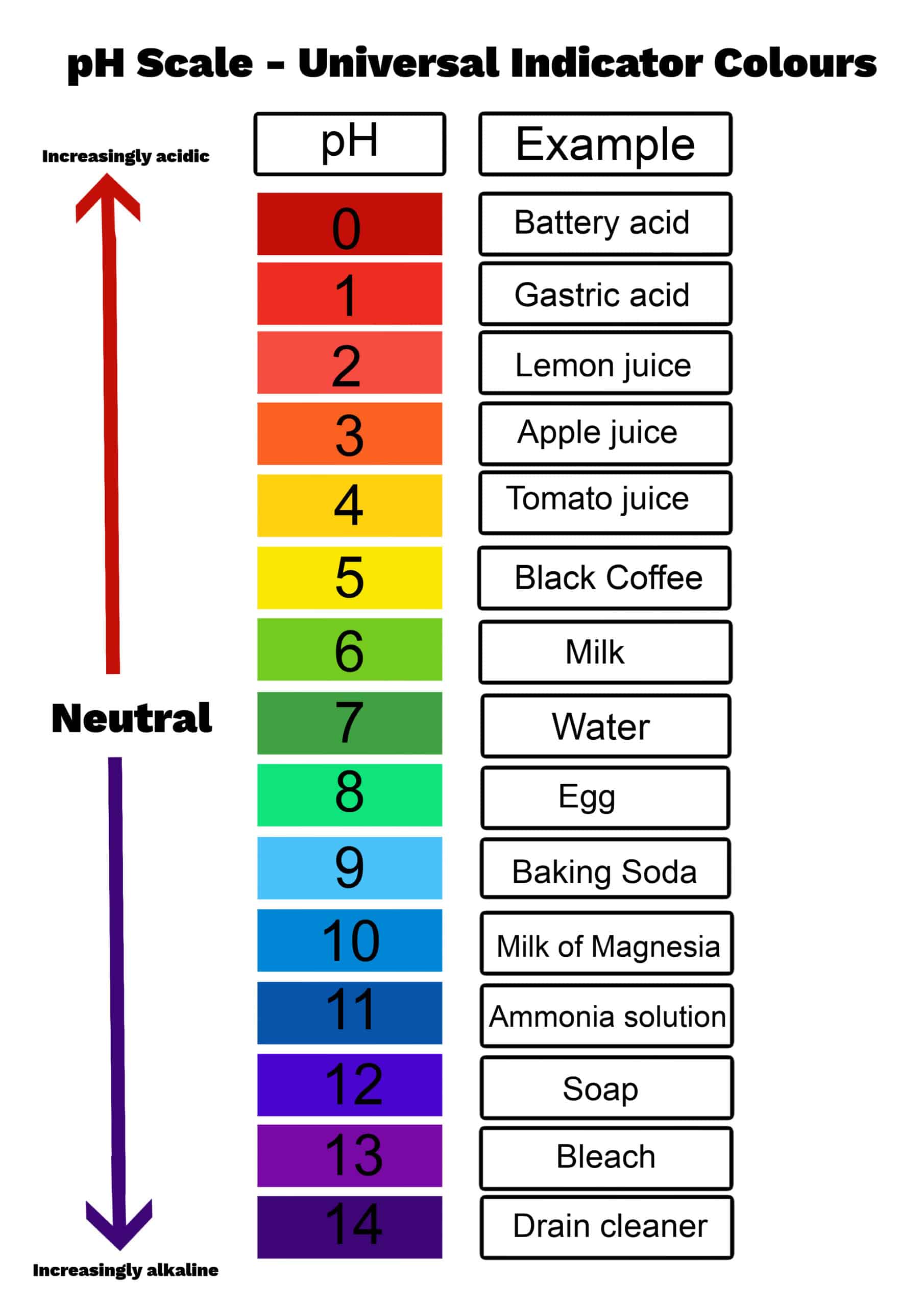
4. CBSE Class 10 Science Notes – pH Scale & Strength
What is pH?
The pH is a measure of hydrogen ion concentration in a solution. It is defined as the negative logarithm of H⁺ ion concentration:
pH Scale (0 to 14)
- pH = 7: Neutral (pure water, NaCl solution)
- pH < 7: Acidic (lower pH = stronger acid)
- pH > 7: Basic/Alkaline (higher pH = stronger base)
- Gastric juice: 1-2 (strongly acidic)
- Lemon juice: 2-3
- Vinegar: 3-4
- Tomato juice: 4-5
- Milk: 6.5-6.8 (slightly acidic)
- Pure water: 7 (neutral)
- Blood: 7.35-7.45 (slightly basic)
- Baking soda: 8-9
- Soap: 9-10
- Household ammonia: 11-12
- Drain cleaner: 13-14 (strongly basic)
Strong vs Weak Acids and Bases
| Type | Strong | Weak |
|---|---|---|
| Acids | HCl, H₂SO₄, HNO₃ (complete dissociation) | CH₃COOH, Citric acid (partial dissociation) |
| Bases | NaOH, KOH, Ca(OH)₂ (complete dissociation) | NH₄OH, Mg(OH)₂ (partial dissociation) |
Importance of pH in Daily Life
1. pH in Our Digestive System
The stomach produces hydrochloric acid (pH 1-2) to digest food and kill bacteria. If excess acid is produced, it causes acidity and indigestion. Antacids (milk of magnesia, baking soda) are used to neutralize this excess acid.
2. pH and Tooth Decay
Bacteria in mouth produce acids by degradation of sugar and food particles. When pH in mouth drops below 5.5, tooth enamel (calcium phosphate) starts corroding. Toothpastes are basic to neutralize this acid.
3. pH of Soil
Plants require specific pH range for healthy growth:
- Most plants: pH 6-7.5
- If soil is acidic: Add slaked lime (Ca(OH)₂) or chalk (CaCO₃)
- If soil is basic: Add organic matter (compost) which releases acids
4. pH of Water Bodies
Aquatic life requires pH 7-8.5. If water becomes too acidic (acid rain, industrial waste), aquatic life is threatened.
5. pH of Blood
Human blood maintains pH 7.35-7.45. Even small changes can be fatal. Buffers in blood maintain this pH.
5. CBSE Class 10 Science Notes – Salts & Chemicals from Common Salt
Definition of Salts
Salts are ionic compounds formed by neutralization reaction between acids and bases. They are composed of positive ions (cations) from bases and negative ions (anions) from acids.
Family of Salts
Salts having the same positive or negative radicals belong to the same family:
- Sodium salts family: NaCl, Na₂SO₄, Na₂CO₃, NaHCO₃
- Chloride salts family: NaCl, KCl, CaCl₂, NH₄Cl
- Sulphate salts family: Na₂SO₄, CuSO₄, ZnSO₄
Types of Salts Based on Strength
| Type | Formation | pH | Examples |
|---|---|---|---|
| Neutral Salt | Strong acid + Strong base | = 7 | NaCl, KNO₃, Na₂SO₄ |
| Acidic Salt | Strong acid + Weak base | < 7 | NH₄Cl, (NH₄)₂SO₄ |
| Basic Salt | Weak acid + Strong base | > 7 | Na₂CO₃, CH₃COONa, NaHCO₃ |
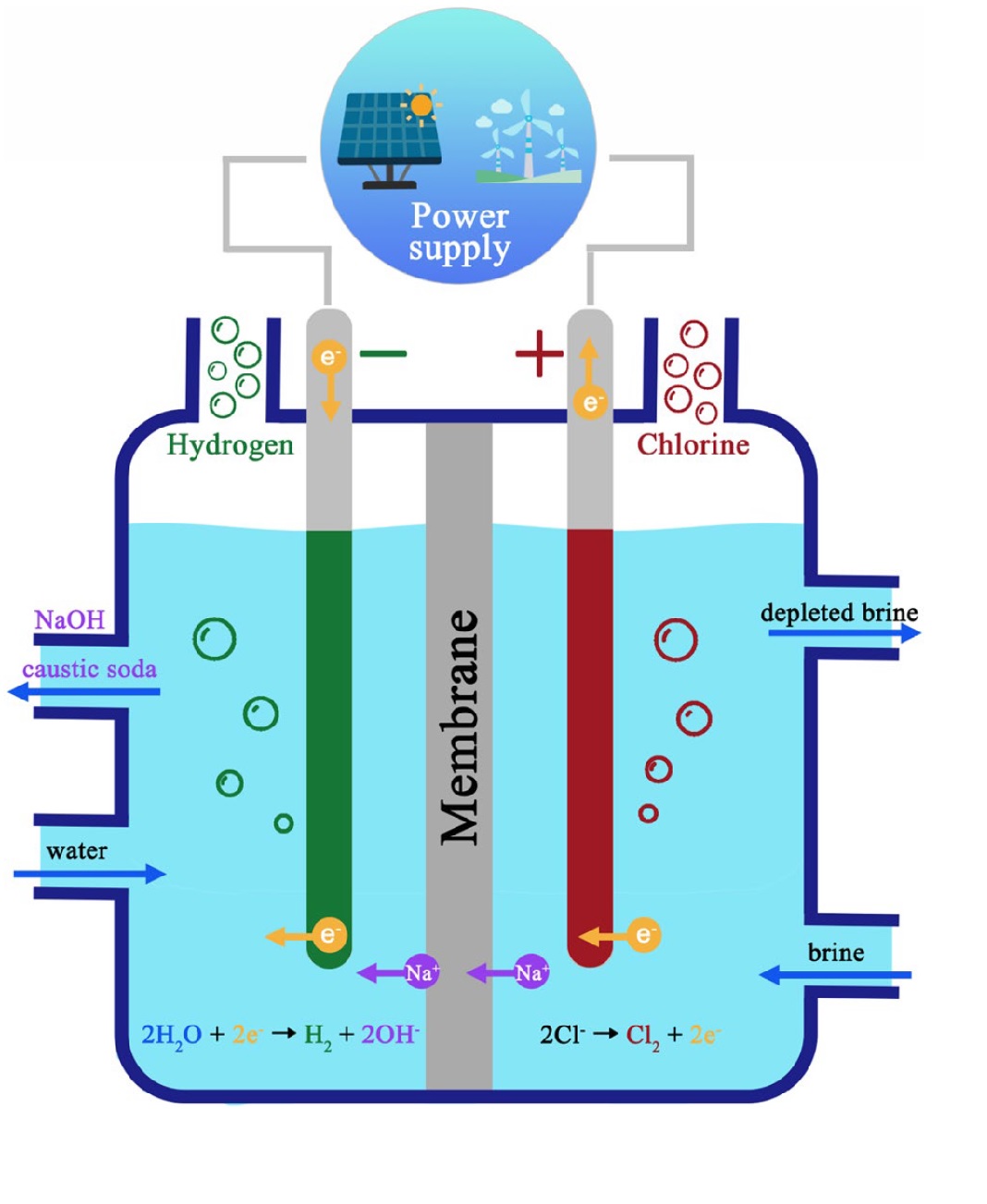
Chlor-Alkali Process
The Chlor-alkali process is an industrial method for producing sodium hydroxide (NaOH), chlorine gas (Cl₂), and hydrogen gas (H₂) by electrolysis of concentrated aqueous sodium chloride solution (brine).
Process Details:
- Brine preparation: Saturated NaCl solution
- Electrolysis: Pass electricity through brine in electrolytic cell
- At anode (positive electrode): Chlorine gas is evolved
2Cl⁻ → Cl₂ + 2e⁻
- At cathode (negative electrode): Hydrogen gas is evolved
2H⁺ + 2e⁻ → H₂
- In solution: Na⁺ and OH⁻ remain, forming NaOH
Products and Their Uses:
| Product | Uses |
|---|---|
| Sodium hydroxide (NaOH) | Soaps, detergents, paper, artificial fibers, petroleum refining |
| Chlorine (Cl₂) | Water treatment, PVC, chloroform, bleaching powder, CFCs |
| Hydrogen (H₂) | Margarine, ammonia synthesis, rocket fuel, hydrogenation of oils |
Important Chemicals from Common Salt
1. Sodium Hydroxide (NaOH) - Caustic Soda
Preparation: Chlor-alkali process (as described above)
Properties: White crystalline solid, hygroscopic, corrosive, soapy touch
Uses: Soap and detergent manufacturing, paper industry, petroleum refining, degreasing metals
2. Bleaching Powder (CaOCl₂)
Preparation: By passing chlorine gas through dry slaked lime
Chemical name: Calcium oxychloride
Uses: Bleaching cotton and linen in textile industry, disinfecting water, making chloroform
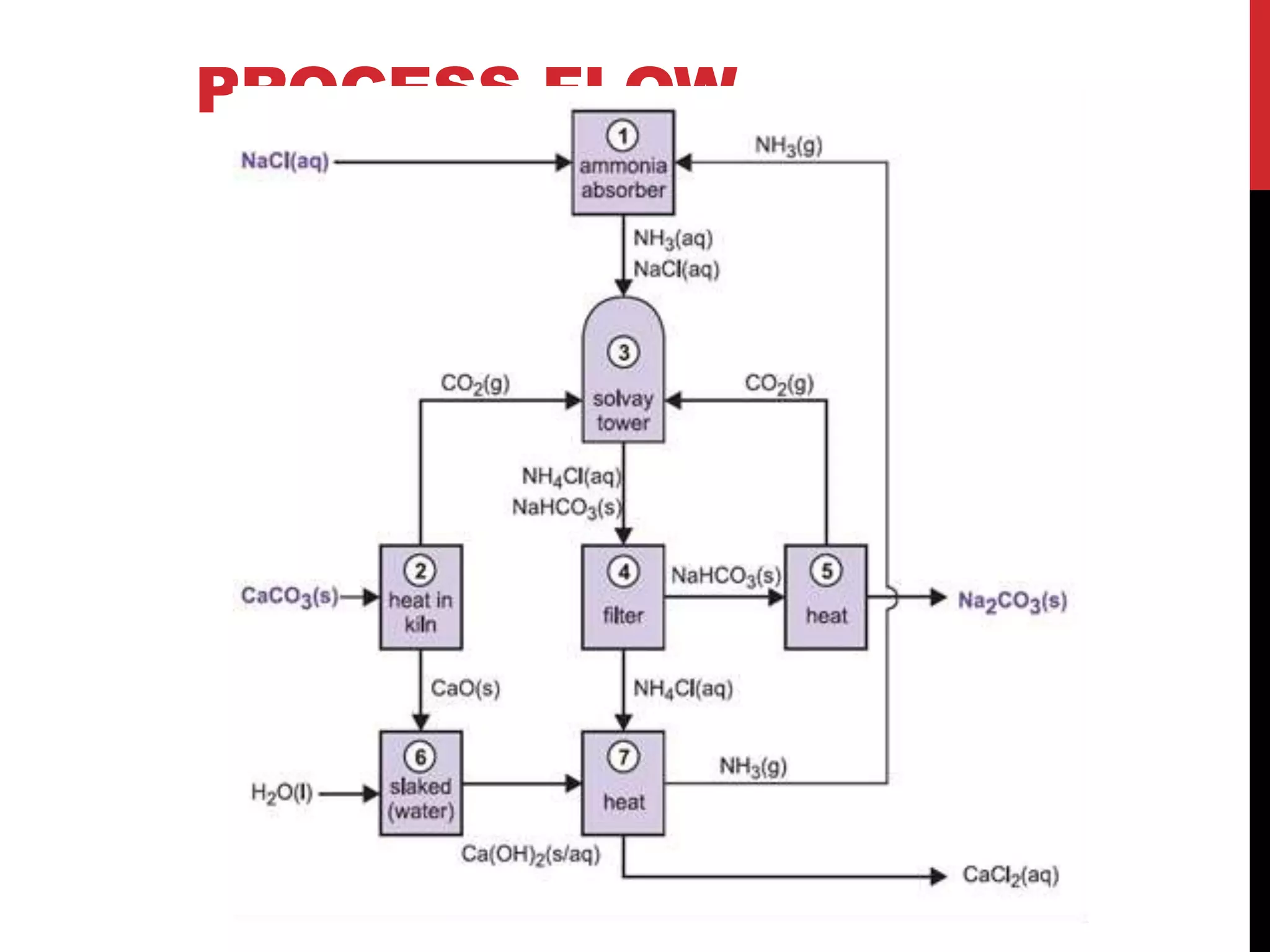
3. Baking Soda (NaHCO₃) - Sodium Bicarbonate
Preparation: By passing CO₂ through ammoniacal brine (Solvay process)
Properties: White crystalline solid, mild non-corrosive base
Uses:
- As baking powder (mixed with tartaric acid)
- Antacid to neutralize stomach acid
- Soda-acid fire extinguishers
- Making soft drinks
4. Washing Soda (Na₂CO₃·10H₂O) - Sodium Carbonate Decahydrate
Preparation: By heating baking soda and then crystallizing
Properties: Transparent crystalline solid, efflorescent (loses water of crystallization)
Uses:
- Manufacturing glass, soap, paper
- Removing permanent hardness of water
- Laundry (cleaning clothes)
Baking Soda = Bread Softener (for cooking)
Washing Soda = Water Softener (for cleaning)
Or: Baking = 1 carbon (NaHCO₃), Washing = 2 carbons (Na₂CO₃)
6. Important Points (Quick Revision)
- pH Scale: 0-6 acidic, 7 neutral, 8-14 basic
- Neutralization: Acid + Base → Salt + Water + Heat
- Strong acids: HCl, H₂SO₄, HNO₃ (complete ionization)
- Weak acids: CH₃COOH, citric acid (partial ionization)
- Chlor-alkali process: Electrolysis of brine → NaOH + Cl₂ + H₂
- Baking soda: NaHCO₃, used in baking powder and antacids
- Washing soda: Na₂CO₃·10H₂O, used in glass and soap industry
- Bleaching powder: CaOCl₂, used for bleaching and disinfection
7. Solved Examples (CBSE Pattern)
Example 1: pH-Based Question (2 Marks)
Question: Five solutions A, B, C, D and E showed pH values of 4, 1, 11, 7 and 9 respectively when tested with universal indicator. Identify:
a) Neutral solution
b) Strongly alkaline solution
c) Strongly acidic solution
d) Weakly acidic solution
e) Weakly alkaline solution
Solution:
- a) D (pH = 7) - Neutral
- b) C (pH = 11) - Strongly alkaline
- c) B (pH = 1) - Strongly acidic
- d) A (pH = 4) - Weakly acidic
- e) E (pH = 9) - Weakly alkaline
Example 2: Reaction with Metal (2 Marks)
Question: A metal compound reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride.
Solution:
The gas that extinguishes candle is CO₂. The metal compound is calcium carbonate (CaCO₃).
Example 3: Identify Acid/Base (3 Marks)
Question: How would you distinguish between baking soda and washing soda using a simple test?
Solution:
Heating Test:
- Baking soda (NaHCO₃): On heating, it decomposes giving off CO₂ gas (turns lime water milky) and water vapor
- Washing soda (Na₂CO₃·10H₂O): On heating, it loses water of crystallization (efflorescence) but no gas evolution
Example 4: Equation-Based Question (3 Marks)
Question: Write balanced chemical equations for:
a) Chlorine gas is passed through dry slaked lime
b) Sodium hydroxide solution reacts with hydrochloric acid
c) Zinc metal reacts with sodium hydroxide
Answers:
Example 5: 5-Mark Conceptual Question
Question: a) What is chlor-alkali process? Write the chemical equation.
b) Name the three products obtained and give one use of each.
c) Why is this process named so?
a) Chlor-alkali process: Electrolysis of concentrated aqueous sodium chloride (brine) to produce sodium hydroxide, chlorine gas and hydrogen gas.
b) Products and uses:
- Sodium hydroxide (NaOH): Making soap and detergents
- Chlorine (Cl₂): Water treatment, PVC production
- Hydrogen (H₂): Hydrogenation of vegetable oils
c) Naming: Named after products - Chlorine and Alkali (NaOH)
Example 6: Case-Study Based Question
Passage: A student observed that fresh milk has pH 6. When it changes into curd, the pH decreases. The milkman adds a small amount of baking soda to fresh milk to increase its pH to slightly alkaline.
Questions: a) Why does pH decrease when milk changes to curd? b) Why does the milkman add baking soda? c) Why does this milk take longer time to set as curd?
a) Milk contains lactic acid bacteria which convert lactose into lactic acid. Acid decreases pH.
b) To shift pH to alkaline so that milk does not set as curd quickly (preservation).
c) The added base (baking soda) neutralizes the lactic acid produced, so more time is needed to produce sufficient acid for curdling.
8. Smart Tricks & Memory Aids
Acid = Angry (low pH, 0-6)
Base = Big heart (high pH, 8-14)
Neutral = Nice (pH 7)
Or remember: 0-6 ACID, 7 NEUTRAL, 8-14 BASE
- Sour taste + Blue litmus turns Red = ACID
- Bitter taste + Soapy feel + Red litmus turns Blue = BASE
- Remember: Blue→Red = Boy→Rude (Acid)
| Baking Soda | Washing Soda |
|---|---|
| NaHCO₃ | Na₂CO₃·10H₂O |
| 1 Na, 1 C | 2 Na, 1 C |
| For cooking/baking | For cleaning/washing |
| Heating → CO₂ released | Heating → Water lost |
Chlorine + Alkali (NaOH) + Hydrogen
Remember: CAH - Chlorine, Alkali, Hydrogen
Or: Chlorine at Anode, Hydrogen at Cathode
- Always write word equation before chemical equation
- Include state symbols (s, l, g, aq) for full marks
- For pH questions: Mention acidic (<7), basic (>7), neutral (=7)
- For salt preparation: Write balanced equation + conditions
- Remember 3 products of chlor-alkali process
- For identification tests: Write observation + conclusion
9. Visual Learning – Diagrams
10. Most Important Board Questions
1 Mark Questions
2-3 Mark Questions
4-5 Mark Questions
11. Common Mistakes to Avoid
| Mistake | Correction |
|---|---|
| Confusing strong and weak acids | Strong acids completely dissociate (HCl, H₂SO₄); Weak acids partially dissociate (CH₃COOH) |
| Writing incorrect chemical formulas | Baking soda = NaHCO₃; Washing soda = Na₂CO₃·10H₂O; Bleaching powder = CaOCl₂ |
| Misunderstanding pH scale | Lower pH = stronger acid; Higher pH = stronger base; pH 7 = neutral |
| Forgetting state symbols | Always include (s), (l), (g), (aq) in final equations |
| Wrong products in chlor-alkali | Remember: NaOH (solution), Cl₂ (anode), H₂ (cathode) |
12. Practice Section
Multiple Choice Questions (MCQs)
Q1. Which of the following has the highest pH value?
- Lemon juice
- Vinegar
- Milk
- Soap solution
Q2. The product of chlor-alkali process are:
- NaCl, H₂, O₂
- NaOH, Cl₂, H₂
- Na₂CO₃, H₂O, CO₂
- NaHCO₃, NH₄Cl
Q3. Which salt is used for removing permanent hardness of water?
- Baking soda
- Washing soda
- Bleaching powder
- Common salt
Assertion-Reason Question
Assertion (A): Toothpaste is basic in nature.
Reason (R): Bacteria in mouth produce acids which cause tooth decay.
Answer: Both A and R are true and R is the correct explanation of A.
Explanation: Bacteria produce acids (pH < 5.5) causing tooth decay. Basic toothpaste neutralizes this acid.
13. Frequently Asked Questions (FAQ)
14. Conclusion
Master Acids, Bases and Salts for Board Success!
These comprehensive CBSE Class 10 Notes for "Acids, Bases and Salts" cover all essential concepts, pH scale, salt preparation, and solved examples to help you excel in your board examinations.
Key Takeaways:
- Understand properties and reactions of acids and bases
- Master pH scale and its importance in daily life
- Remember chlor-alkali process and its three products
- Practice chemical equations with state symbols
- Distinguish between baking soda and washing soda
Next Chapter: Metals and Non-metals – CBSE Class 10 Notes
Best of luck for your CBSE Class 10 Board Examinations!
These CBSE Class 10 Science Notes are prepared by experienced Chemistry faculty following the latest NCERT curriculum.

