
1. Introduction to Carbon Chemistry
Welcome to the most scoring chapter of your CBSE Class 10 Science Notes. Carbon and its Compounds (Chapter 4) is a fundamental topic that bridges basic chemistry with real-world applications. This chapter carries approximately 5-7 marks in the CBSE Board Examination, making it crucial for your overall Science score.
Why is Carbon Special?
Carbon is unique among all elements due to two extraordinary properties:
- Tetravalency: Carbon has 4 valence electrons, enabling it to form 4 stable covalent bonds
- Catenation: Carbon atoms can link with each other to form long chains, branches, and rings
These CBSE Class 10 Notes are strictly aligned with the latest NCERT syllabus (2024-25) and designed to help you master every concept from covalent bonding to soap chemistry.
2. Chapter Overview: CBSE Class 10 Science Notes
Key Topics Covered:
- Covalent Bonding – Sharing of electrons between atoms
- Electron Dot Structures – Lewis structures for carbon compounds
- Allotropes of Carbon – Diamond, Graphite, and Fullerenes
- Hydrocarbons – Saturated (Alkanes) and Unsaturated (Alkenes/Alkynes)
- Homologous Series – Systematic classification of organic compounds
- Functional Groups – Characteristic groups determining chemical properties
- Chemical Properties – Combustion, oxidation, addition, and substitution reactions
- Important Compounds – Ethanol and Ethanoic Acid
- Soaps and Detergents – Cleansing action and micelle formation
3. Key Concepts (NCERT Aligned)
3.1 Covalent Bonding in Carbon
Definition: A covalent bond is formed by the mutual sharing of electrons between two atoms to achieve the stable electronic configuration of the nearest noble gas.
Why Carbon Forms Covalent Bonds:
- Carbon has 4 valence electrons (electronic configuration: 2, 4)
- It cannot lose 4 electrons (requires too much energy)
- It cannot gain 4 electrons (nucleus cannot hold 8 extra electrons)
- Solution: Shares 4 electrons with other atoms
Types of Covalent Bonds:
- Single Bond (C-C): Sharing of 1 pair of electrons (Alkanes)
- Double Bond (C=C): Sharing of 2 pairs of electrons (Alkenes)
- Triple Bond (C≡C): Sharing of 3 pairs of electrons (Alkynes)
Electron Dot Structures:
| Compound | Structure | Bond Type |
|---|---|---|
| Methane (CH4) | H H:C:H H |
4 single C-H bonds |
| Ethane (C2H6) | H H H:C:C:H H H |
1 C-C single bond, 6 C-H bonds |
| Ethene (C2H4) | H H H:C::C:H |
1 C=C double bond |
| Ethyne (C2H2) | H:C:::C:H | 1 C≡C triple bond |
3.2 Allotropes of Carbon
Definition: Allotropes are different forms of the same element with distinct physical properties due to different structural arrangements.
1. Diamond:
- Each carbon bonded to 4 others in tetrahedral structure
- Hardest natural substance
- Bad conductor of electricity (no free electrons)
- Used in cutting tools and jewelry
2. Graphite:
- Each carbon bonded to 3 others in hexagonal layers
- Soft and slippery (layers slide over each other)
- Good conductor (free electrons between layers)
- Used as lubricant and in pencil leads
3. Fullerenes:
- Hollow cage-like structures (C60, C70)
- Carbon atoms arranged in football-like shape
- Used in drug delivery and nanotechnology

3.3 Hydrocarbons
Definition: Organic compounds containing only carbon and hydrogen.
Saturated Hydrocarbons (Alkanes)
- General Formula: CnH2n+2
- Characteristics: Only single covalent bonds (C-C)
- Examples:
- Methane (CH4) - n=1
- Ethane (C2H6) - n=2
- Propane (C3H8) - n=3
Unsaturated Hydrocarbons
Alkenes:
- General Formula: CnH2n
- Characteristics: Contains at least one C=C double bond
- Examples: Ethene (C2H4), Propene (C3H6)
Alkynes:
- General Formula: CnH2n-2
- Characteristics: Contains at least one C≡C triple bond
- Examples: Ethyne (C2H2), Propyne (C3H4)
3.4 Homologous Series
Definition: A series of organic compounds with the same functional group and similar chemical properties, where each successive member differs by a -CH2 group (methylene group) and 14 u (atomic mass unit).
Characteristics:
- Same general formula
- Similar chemical properties
- Gradual change in physical properties (boiling point increases with carbon number)
- Can be represented by same general formula

| Series | General Formula | Functional Group | Example |
|---|---|---|---|
| Alkanes | CnH2n+2 | C-C (single) | CH4 (Methane) |
| Alkenes | CnH2n | C=C (double) | C2H4 (Ethene) |
| Alkynes | CnH2n-2 | C≡C (triple) | C2H2 (Ethyne) |
| Alcohols | CnH2n+1OH | -OH | CH3OH (Methanol) |
| Carboxylic Acids | CnH2n+1COOH | -COOH | CH3COOH (Ethanoic acid) |
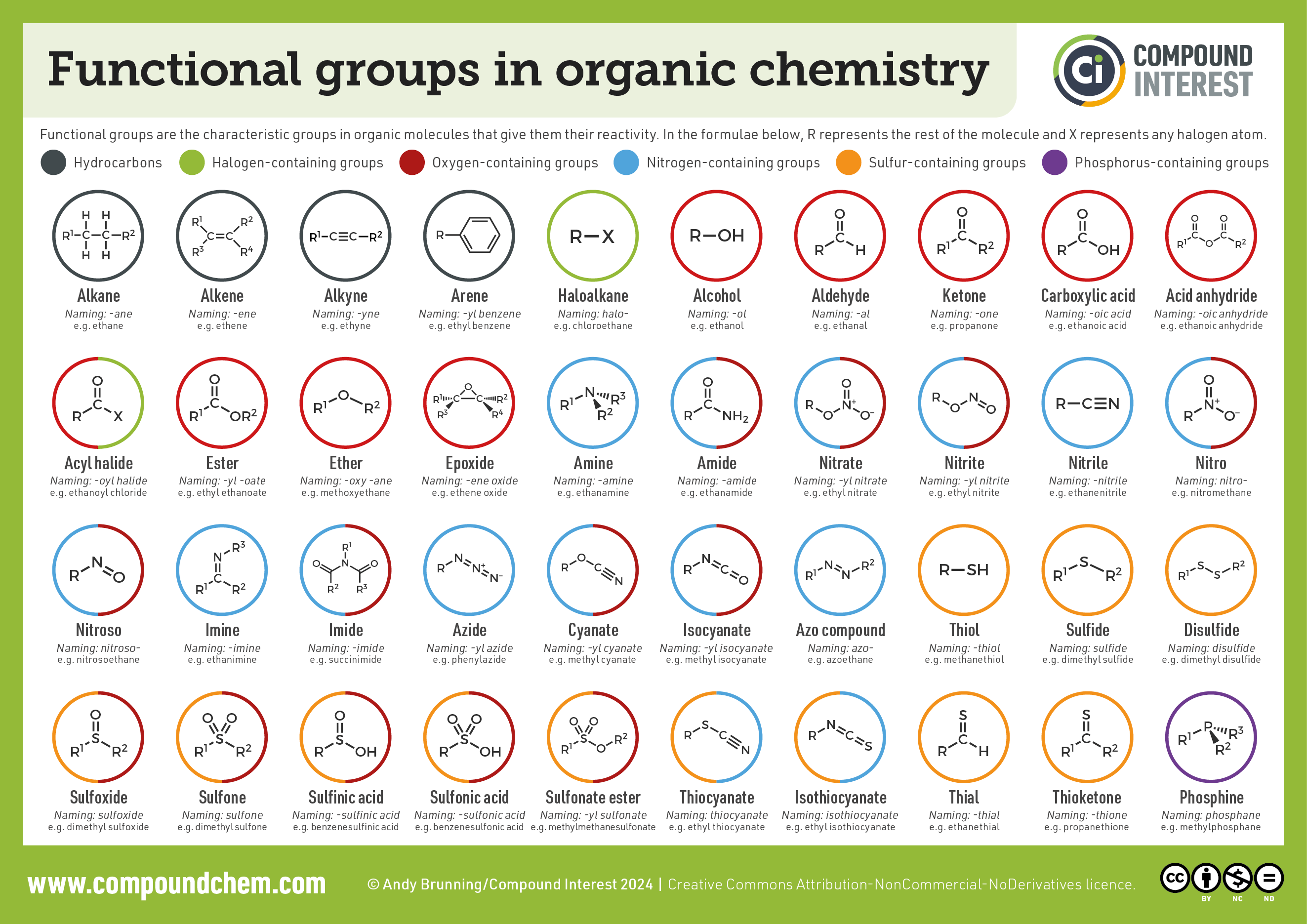
3.5 Functional Groups
Definition: An atom or group of atoms that determines the characteristic chemical properties of an organic compound.
| Functional Group | Name | Class of Compound | Example |
|---|---|---|---|
| -OH | Hydroxyl | Alcohol | Ethanol (C2H5OH) |
| -COOH | Carboxyl | Carboxylic Acid | Ethanoic acid (CH3COOH) |
| -CHO | Aldehyde | Aldehyde | Methanal (HCHO) |
| -CO- | Carbonyl | Ketone | Propanone (CH3COCH3) |
| -X (F, Cl, Br, I) | Halogen | Haloalkane | Chloromethane (CH3Cl) |
| -NH2 | Amino | Amine | Methanamine (CH3NH2) |
3.6 Chemical Properties of Carbon Compounds
1. Combustion
Carbon compounds burn in oxygen to produce CO2, water, and heat/light.
General Reaction:
Examples:
- Methane: CH4 + 2O2 → CO2 + 2H2O + Heat + Light
- Ethane: 2C2H6 + 7O2 → 4CO2 + 6H2O + Heat
2. Oxidation
Addition of oxygen or removal of hydrogen.
Ethanol to Ethanoic Acid:
Oxidizing Agents: Alkaline KMnO4 or Acidified K2Cr2O7 (they get reduced)
3. Addition Reaction
Unsaturated hydrocarbons add atoms at double/triple bond positions.
Hydrogenation of Ethene:
4. Substitution Reaction
Saturated hydrocarbons undergo substitution where hydrogen is replaced by another atom/group.
Chlorination of Methane:
Mechanism: Free radical mechanism initiated by UV light/sunlight
3.7 Ethanol and Ethanoic Acid
Ethanol (C2H5OH) - Ethyl Alcohol
- Preparation: Fermentation of sugars by yeast
C6H12O6 →[Yeast] 2C2H5OH + 2CO2
- Properties:
- Colorless liquid, pleasant smell
- Miscible with water (forms H-bonds)
- Dehydration: With conc. H2SO4 at 443K forms ethene
- Reaction with Sodium: 2C2H5OH + 2Na → 2C2H5ONa + H2↑
Ethanoic Acid (CH3COOH) - Acetic Acid
- Properties:
- Weak acid (5-8% solution = vinegar)
- Esterification: Reacts with alcohol to form esters (sweet-smelling compounds)
CH3COOH + C2H5OH →[Conc. H2SO4] CH3COOC2H5 + H2O
- Reaction with Base: Neutralization reaction
CH3COOH + NaOH → CH3COONa + H2O
- Reaction with Carbonates:
2CH3COOH + Na2CO3 → 2CH3COONa + H2O + CO2↑
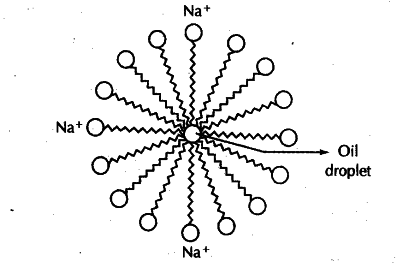
3.8 Soaps and Detergents
Soap: Sodium or potassium salts of long-chain carboxylic acids (fatty acids)
- General Formula: R-COONa (R = long alkyl chain, C12-C18)
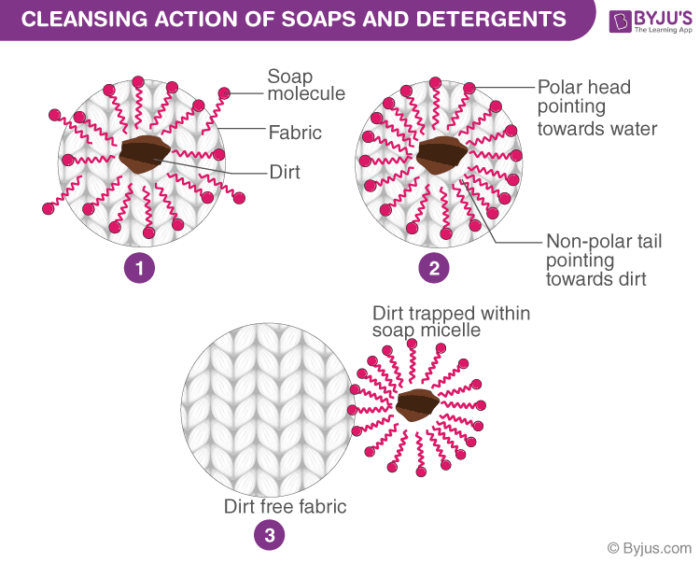
Cleansing Action of Soap:
- Soap molecules have hydrophobic tail (water-repelling, dissolves in oil) and hydrophilic head (water-attracting, dissolves in water)
- In water, soap forms micelles - spherical clusters with hydrophobic tails inward (trapping dirt/oil) and hydrophilic heads outward
- Micelles remain suspended in water due to ion-ion repulsion
- Rinsing washes away micelles with trapped dirt

Difference between Soap and Detergent:
| Feature | Soap | Detergent |
|---|---|---|
| Source | Natural (vegetable oils/animal fats) | Synthetic (petrochemicals) |
| Action in hard water | Forms insoluble scum (Ca/Mg salts) | Works effectively |
| Biodegradability | Biodegradable | Some are non-biodegradable |
| pH | Basic | Can be neutral/acidic |
4. Important Formulas and Structures
Hydrocarbon General Formulas
Alkanes (Saturated): CnH2n+2
Alkenes (Unsaturated): CnH2n
Alkynes (Unsaturated): CnH2n-2
Functional Group Quick Chart
Alcohol: -OH (Suffix: -ol)
Aldehyde: -CHO (Suffix: -al)
Ketone: -CO- (Suffix: -one)
Carboxylic Acid: -COOH (Suffix: -oic acid)
Halogen: -X (Cl, Br) (Prefix: chloro-, bromo-)
IUPAC Naming Steps:
- Identify longest carbon chain (root word: meth-, eth-, prop-, but-, pent-)
- Identify functional group (suffix/prefix)
- Number the chain from end nearest to functional group
- Name substituents with position numbers
- Arrange alphabetically
Example: CH3-CH2-CH2-OH
- 3 carbons = prop-
- -OH group = -anol
- Name: Propan-1-ol (or 1-Propanol)
5. Solved Examples (CBSE Pattern)
Question: Draw the electron dot structure of ethene (C2H4).
- Ethene has 2 carbon atoms and 4 hydrogen atoms
- Each carbon forms 3 bonds (1 with other C, 2 with H atoms)
- Carbon atoms share 2 pairs of electrons (double bond)
- Total valence electrons: (2×4) + (4×1) = 12 electrons = 6 pairs
Structure:
H H
\ /
C=C
/ \
H H
Answer: Ethene has one C=C double bond and four C-H single bonds.
Question: Identify the functional groups in:
(a) CH3COCH3
(b) CH3CH2CHO
(c) C2H5OH
(a) CH3COCH3: Contains -CO- group → Ketone (Propanone/Acetone)
(b) CH3CH2CHO: Contains -CHO group → Aldehyde (Propanal)
(c) C2H5OH: Contains -OH group → Alcohol (Ethanol)
Question: Write balanced chemical equations for:
(a) Combustion of propane
(b) Hydrogenation of ethene
(a) Combustion of Propane (C3H8):
(b) Hydrogenation of Ethene:
Question: Why are carbon and its compounds used as fuels?
Carbon compounds are used as fuels because:
1. High calorific value: They produce large amount of heat per unit mass
2. Complete combustion: Saturated hydrocarbons burn completely producing only CO2 and H2O
3. Easy availability: Fossil fuels (coal, petroleum, natural gas) are rich in carbon compounds
4. Controlled combustion: Can be easily ignited and controlled for various applications
Question: A student observed that when ethanol is heated with alkaline KMnO4, the pink color disappears and a new compound with acidic properties is formed. Identify the new compound and write the reaction. Why does the color disappear?
- New Compound: Ethanoic acid (CH3COOH)
- Reaction:
- Explanation: Alkaline KMnO4 (pink) is an oxidizing agent that oxidizes ethanol to ethanoic acid. During this process, KMnO4 gets reduced (loses oxygen), causing the pink color to disappear.
6. Smart Tricks and Memory Aids
Functional Group Memory Trick
Remember the sequence: "Alcohols Are Cool, Kings Care About Health"
- Alcohol (-OH)
- Aldehyde (-CHO)
- Carboxylic acid (-COOH)
- Ketone (-CO-)
- Halogen (-X)
Homologous Series Shortcut
- Difference is always CH2 (14 u)
- Molecular mass increases by 14 between consecutive members
- Boiling point increases with increasing carbon number (due to increased van der Waals forces)
Reaction Identification Shortcut
- Addition Reaction: Only unsaturated compounds (alkenes/alkynes) + atoms added
- Substitution Reaction: Only saturated compounds (alkanes) + atom replaces H
- Combustion: All carbon compounds + O2 → CO2 + H2O
- Oxidation: Alcohol → Acid (gain of O/loss of H)
IUPAC Naming Quick Trick
"Meth-Eth-Prop-But-Pent-Hex" (1-6 carbons)
Count carbons → Add functional group suffix → Number from nearest functional group
Board Exam Writing Strategy
- Draw electron dot structures with clear dots and crosses
- Always balance chemical equations
- Write conditions (temperature, catalyst, pressure) above/below arrow
- For mechanism questions, use bullet points for clarity
7. Visual Learning: Important Diagrams
Diagram 1: Electron Dot Structure of Methane

Key Points to Label:
- Central carbon atom with 4 valence electrons
- Four hydrogen atoms each sharing 1 electron
- Four C-H single covalent bonds
Diagram 2: Diamond vs Graphite Structure

| Property | Diamond | Graphite |
|---|---|---|
| Bonding | 4 bonds (tetrahedral) | 3 bonds (planar hexagonal) |
| Hardness | Very hard | Soft/slippery |
| Conductor | Insulator | Conductor |
| Uses | Cutting, jewelry | Lubricant, pencils |
Diagram 3: Homologous Series Chart
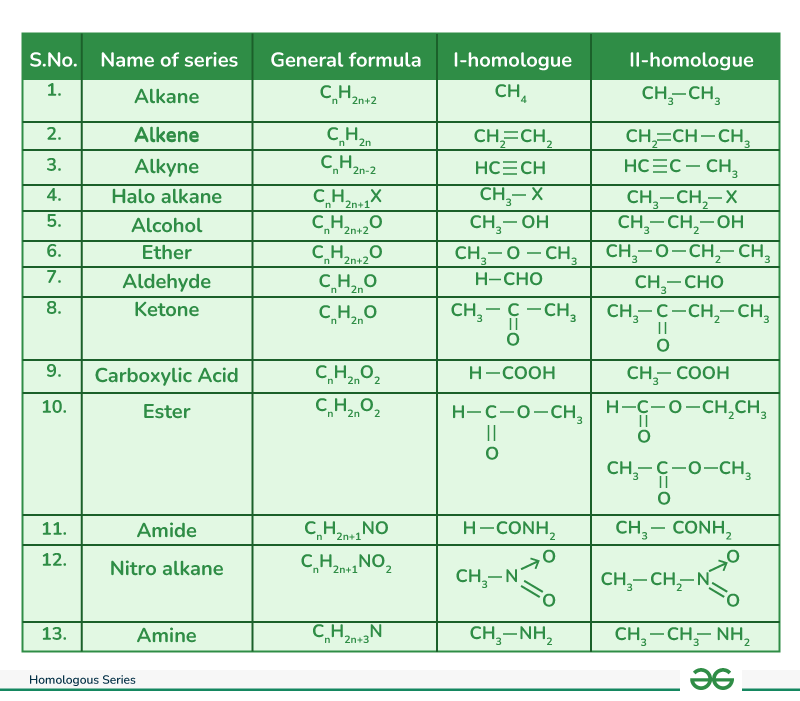
Diagram 4: Functional Groups Table
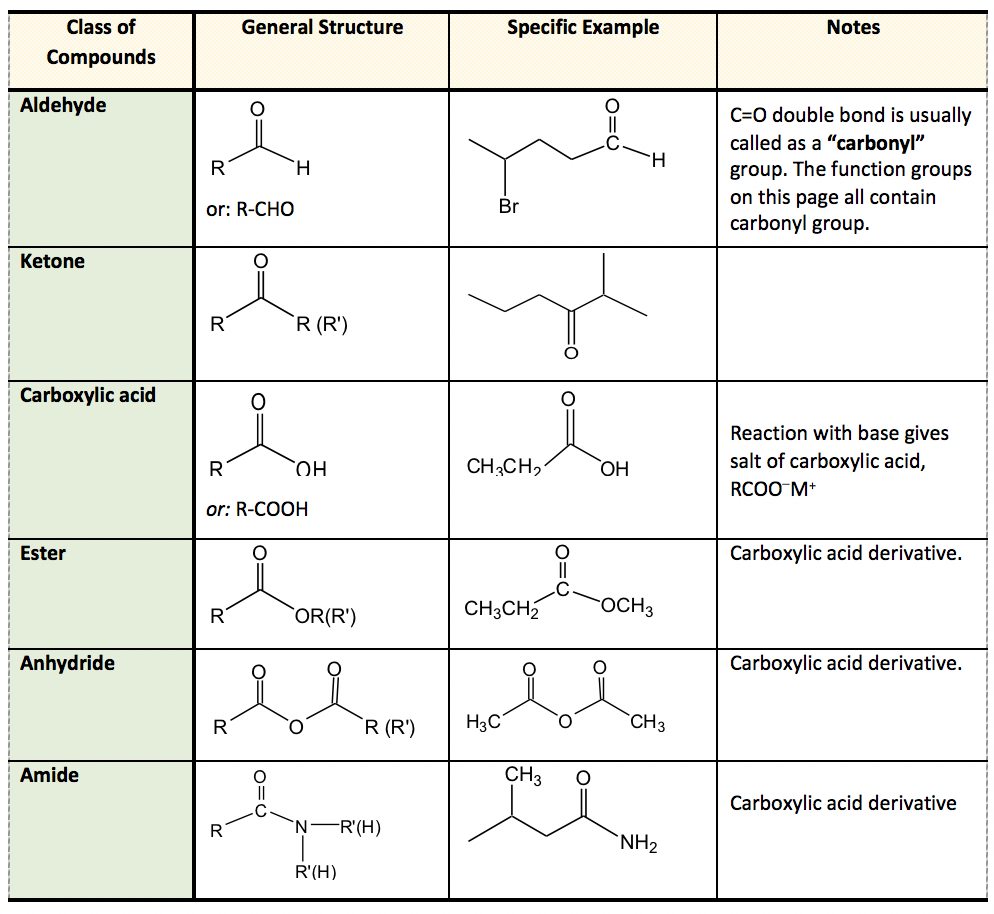
Diagram 5: Micelle Formation
8. Most Important Board Questions
1 Mark Questions (Very Short Answer)
Ans: Carbon
Ans: CnH2n
Ans: Hydroxyl group (-OH)
Ans: Carbon has 4 valence electrons; it cannot lose or gain 4 electrons, so it shares electrons to complete its octet.
Ans: Ethanoic acid (Acetic acid)
Ans: Addition of hydrogen to unsaturated hydrocarbons in presence of catalyst (Ni/Pd/Pt).
2-3 Mark Questions (Short Answer)
| Saturated Hydrocarbons | Unsaturated Hydrocarbons |
|---|---|
| Contain only C-C single bonds | Contain C=C or C≡C bonds |
| General formula: CnH2n+2 | General formula: CnH2n or CnH2n-2 |
| Less reactive | More reactive |
| Example: Ethane (C2H6) | Example: Ethene (C2H4) |
Ethyne has triple bond between carbons:
H:C:::C:H
Each carbon shares 3 electrons with the other carbon (triple bond) and 1 electron with hydrogen.
Hard water contains Ca2+ and Mg2+ ions. Soap reacts with these ions to form insoluble calcium/magnesium salts (scum/precipitate), which does not clean clothes effectively and wastes soap.
(a) Combustion of methane
(b) Oxidation of ethanol
(a) CH4 + 2O2 → CO2 + 2H2O + Heat + Light
(b) C2H5OH + 2[O] → CH3COOH + H2O (using alkaline KMnO4)
4-5 Mark Questions (Long Answer/Conceptual)
Soap molecules are sodium/potassium salts of long-chain fatty acids with:
- Hydrophobic tail (water-repelling, dissolves in oil/grease)
- Hydrophilic head (water-attracting, dissolves in water)
Mechanism:
- Soap dissolves in water forming ions
- Hydrophobic tails attach to oil/dirt particles
- Hydrophilic heads remain in water
- Molecules arrange into spherical micelles with dirt trapped inside
- Micelles remain suspended due to ion-ion repulsion
- Rinsing washes away micelles with trapped dirt
Chemical Equation: R-COONa → R-COO- + Na+ (in water)
Definition: A series of organic compounds with the same functional group and similar chemical properties, where successive members differ by -CH2 group (14 u).
Examples:
- Alkanes: CH4, C2H6, C3H8, C4H10...
- Alcohols: CH3OH, C2H5OH, C3H7OH...
Characteristics:
- Same general formula and functional group
- Similar chemical properties
- Gradual change in physical properties (bp/mp increase with molecular mass)
- Consecutive members differ by CH2 group (14 u)
Diamond:
- Each C bonded to 4 others (tetrahedral)
- 3D rigid network structure
- No free electrons → Non-conductor
- Very hard (used for cutting)
Graphite:
- Each C bonded to 3 others (hexagonal layers)
- Layered structure with weak forces between layers
- Free electrons between layers → Good conductor
- Soft and slippery (used as lubricant)
Conclusion: Different arrangements of same carbon atoms create vastly different properties.
Chemical Equation Questions
- CH4 + 2O2 → CO2 + 2H2O
- C2H4 + H2 → C2H6 (Hydrogenation)
- CH4 + Cl2 → CH3Cl + HCl (Substitution)
- C2H5OH + 3O2 → 2CO2 + 3H2O
- CH3COOH + NaOH → CH3COONa + H2O
- CH3COOH + C2H5OH → CH3COOC2H5 + H2O (Esterification)
- 2CH3COOH + Na2CO3 → 2CH3COONa + H2O + CO2
Case-Study Based Question (CBSE Pattern)
A student performed an experiment where Compound X (C2H4O2) reacted with sodium carbonate to produce brisk effervescence of gas Y. When gas Y was passed through lime water, it turned milky. Compound X also reacted with ethanol in presence of conc. H2SO4 to produce sweet-smelling Compound Z.
(a) Identify Compound X, Gas Y, and Compound Z.
- X = Ethanoic acid (CH3COOH)
- Y = Carbon dioxide (CO2)
- Z = Ethyl ethanoate (Ester, CH3COOC2H5)
(b) Write chemical equations for both reactions.
1. 2CH3COOH + Na2CO3 → 2CH3COONa + H2O + CO2↑
2. CH3COOH + C2H5OH → CH3COOC2H5 + H2O (Esterification)
(c) What is the role of conc. H2SO4 in the second reaction?
Conc. H2SO4 acts as a dehydrating agent and catalyst. It absorbs water produced in the reaction, shifting equilibrium towards ester formation (Le Chatelier's principle).
9. Common Mistakes to Avoid
Mistake 1: Incorrect Functional Group Writing
- Wrong: Writing -COH for aldehyde
- Correct: Aldehyde is -CHO (Carbon bonded to both H and O)
Mistake 2: Unbalanced Chemical Equations
- Always check atom balance on both sides
- Remember to balance O2 molecules in combustion reactions
Mistake 3: Confusing Addition vs Substitution
- Addition: Only for unsaturated compounds (C=C or C≡C bonds break, atoms add)
- Substitution: Only for saturated compounds (H replaced by another atom)
Mistake 4: IUPAC Naming Errors
- Number chain from end nearest to functional group
- Use lowest possible numbers for substituents
- Don't forget to mention position numbers for functional groups
Mistake 5: Electron Dot Structure Mistakes
- Carbon must have exactly 4 bonds (8 electrons in outer shell)
- Hydrogen can have only 1 bond (2 electrons)
- Show shared pairs clearly between atoms
Mistake 6: Forgetting Reaction Conditions
- Hydrogenation: Ni/Pd/Pt catalyst
- Combustion: Heat/spark
- Substitution: UV light/Sunlight
- Esterification: Conc. H2SO4, heat
10. Practice Section
MCQs (Multiple Choice Questions)
(a) CnH2n+2
(b) CnH2n
(c) CnH2n-2
(d) CnHn
Ans: (c) CnH2n-2
(a) -OH
(b) -CHO
(c) -COOH
(d) -CO-
Ans: (c) -COOH
(a) Combustion
(b) Hydrogenation
(c) Oxidation
(d) Saponification
Ans: (b) Hydrogenation
(a) Diamond
(b) Graphite
(c) Fullerene
(d) Coal
Ans: (b) Graphite
(a) Alcohol
(b) Soap solution
(c) Salt solution
(d) Acid solution
Ans: (b) Soap solution
Assertion-Reason Questions
Reason: Carbon cannot lose or gain four electrons to form ionic bonds.
(a) Both A and R are true, R explains A
(b) Both A and R are true, R does not explain A
(c) A is true, R is false
(d) A is false, R is true
Ans: (a) Both true, R explains A
Reason: Soaps form insoluble precipitates with Ca2+ and Mg2+ ions.
Ans: (a) Both true, R explains A
HOTS (Higher Order Thinking Skills)
In graphite, carbon atoms are arranged in hexagonal layers held by weak van der Waals forces, allowing layers to slide over each other. In diamond, each carbon is bonded to four others in a rigid 3D tetrahedral network, making it extremely hard.
The compound is Propanoic acid (CH3CH2COOH). It contains -COOH group (carboxylic acid) which reacts with Na2CO3 to produce CO2 gas.
11. Frequently Asked Questions (FAQ)
Carbon has atomic number 6 with electronic configuration 2,4. It has 4 valence electrons in its outermost shell. To achieve stable noble gas configuration (octet), it needs 4 more electrons. Since it cannot easily lose or gain 4 electrons, it shares 4 electrons through covalent bonding, making it tetravalent.
A homologous series is a group of organic compounds with the same functional group and similar chemical properties, where each successive member differs by a -CH2 group (methylene group) and 14 atomic mass units. Examples include alkanes, alkenes, and alcohols.
Soaps clean through micelle formation. Soap molecules have hydrophobic tails (attracted to oil/dirt) and hydrophilic heads (attracted to water). In water, they form spherical micelles trapping dirt inside, which are then washed away with water.
Yes, these CBSE Class 10 Science Notes are 100% aligned with the latest NCERT syllabus (2024-25). All concepts, definitions, and chemical equations follow NCERT guidelines strictly to ensure accuracy for board examinations.
Carbon and its Compounds carries approximately 5-7 marks in the CBSE Class 10 Science Board Examination, typically including 1-mark MCQs, 2-mark short answers, and 3-5 mark long answer questions.
Use the mnemonic "Alcohols Are Cool, Kings Care About Health" for Alcohol, Aldehyde, Carboxylic acid, Ketone, and Halogen. Practice writing their structures daily and associate them with common examples (ethanol in alcohol, vinegar for carboxylic acid).
12. Conclusion
Mastering Carbon and its Compounds is essential for scoring well in your CBSE Class 10 Science Board Examination. This chapter forms the foundation for advanced organic chemistry in higher classes.
Key Takeaways:
- Understand covalent bonding and electron dot structures
- Memorize general formulas of hydrocarbons
- Master functional groups and their properties
- Practice writing balanced chemical equations
- Learn IUPAC nomenclature rules
- Understand micelle formation for soaps
Revision Strategy:
- Revise these CBSE Class 10 Notes regularly
- Practice all chemical equations daily
- Draw structures multiple times for muscle memory
- Solve previous year board questions
- Attempt mock tests under exam conditions
These CBSE Class 10 Science Notes for Chapter 4 Carbon and its Compounds are prepared by experienced Chemistry faculty following the latest NCERT curriculum.

